Als and stem cell treatment7/24/2023  
Although one RCT provided low-certainty evidence that BM-MSC may slightly reduce functional impairment measured on the ALSFRS-R after four to six months, this was a small phase II trial that cannot be used to establish efficacy. 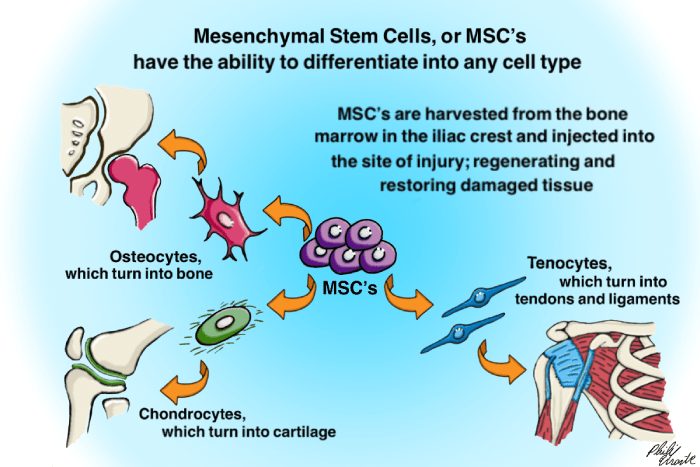
Uncertainties remain as to whether this mode of therapy is capable of restoring muscle function, slowing disease progression, and improving survival in people with ALS/MND. The evidence is up to date as of July 2019.Ĭurrently, there is a lack of high-certainty evidence to guide practice on the use of cell-based therapy to treat ALS/MND. Major goals of future research are to identify the right type and amount of cells to use, and how best to administer them. We urgently need large, well-designed clinical trials to establish whether or not cell-based therapies have a clear clinical benefit in ALS/MND. Based on the very limited evidence available, any benefit is uncertain due to there being only one poorly conducted study and results within the study varies. Based on evidence from this trial, stem cell treatment may slightly reduce decline in motor function at six months, but may not improve breathing or quality of life at four months, or overall survival at six months. The cell implantation procedure was well tolerated. The study provided low-quality evidence that stem cells obtained from people's own bone marrow (the cells in the centre of bone) did not result in significant side effects. They had mild to moderate problems with motor function (ability to perform physical tasks) at the start of the trial (with an average of 35 on the ALS Functional Rating Scale-revised, on which a score of 0 indicates greatest impairment and 48 is normal function). The people taking part in the trial had an average time since symptom onset of about two years. One study, which included 64 people with ALS/MND, provided data. 
Both studies were funded by stem cell companies. One study was not fully published and did not provide numerical data. They found two completed RCTs that assessed the effects of cell-based therapy over a six-month follow-up period. We undertook this review to assess the RCT evidence now becoming available.Ĭochrane review authors searched medical databases for clinical trials. There is currently no approved cell-based therapy for ALS/MND. Limited data from non-RCTs involving a small number of people with ALS/MND and a short follow-up period suggested that cell-based therapy may slow disease progression. Studies with no untreated group for comparison and small clinical trials have found no clinical benefits. Well-performed RCTs provide the best evidence. In RCTs, one group receives the test treatment, and the other, 'control' group has an alternative treatment, a dummy treatment (placebo) or no treatment. Randomised controlled trials (RCTs) provide the most reliable evidence. Previous reviews supported the use of cell-based therapy as a potential means of delaying the disease course in ALS/MND, but these were mainly based on preclinical animal models. Stem cell therapy aims to provide new motor neurons, which may help stop or slow down disease progression in people with ALS/MND. Various types of cell-based therapies have been tried in ALS/MND, including stem cell therapy. Current treatment approaches largely focus on relieving symptoms to improve the quality of life of those affected.Ĭell-based therapy can be defined as injection of cellular material into a person to treat disease. Weakness of muscles used in breathing often leads to death. Half of people with ALS/MND die within three years of their first symptoms. A person with ALS/MND has difficulty moving, swallowing, chewing and speaking, which become worse over time. How effective and safe is cell-based therapy in people with ALS/MND, when we compare it with an inactive treatment or no treatment?Īmyotrophic lateral sclerosis (ALS also known as motor neuron disease or MND) is a condition in which nerves in the brain and spinal cord that control movement (motor neurons) stop working. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
